Thinking Beyond the Thermometer — Smart Sample Transport for ISO 15189
Ensuring that samples are transported under the right conditions is essential for both quality and compliance in modern laboratories. ISO 15189:2022 makes these requirements even clearer and more demanding, going beyond simply recording temperatures to emphasize total traceability and actionable data throughout the transport process.
Why Traditional Methods Fall Short
Most laboratories are familiar with using thermometers and basic data loggers, recorded manually or on paper. While this approach is straightforward and cheap, it introduces significant gaps:
- Data is often delayed and fragmented, making it hard to respond to temperature deviations in real time.
- Paper logs and basic devices are prone to error, loss, or inconsistent documentation, especially during hectic periods or audits.
- Critical issues, such as brief temperature spikes or accidental rough handling (shock events), may go unrecorded, compromising both quality and compliance.
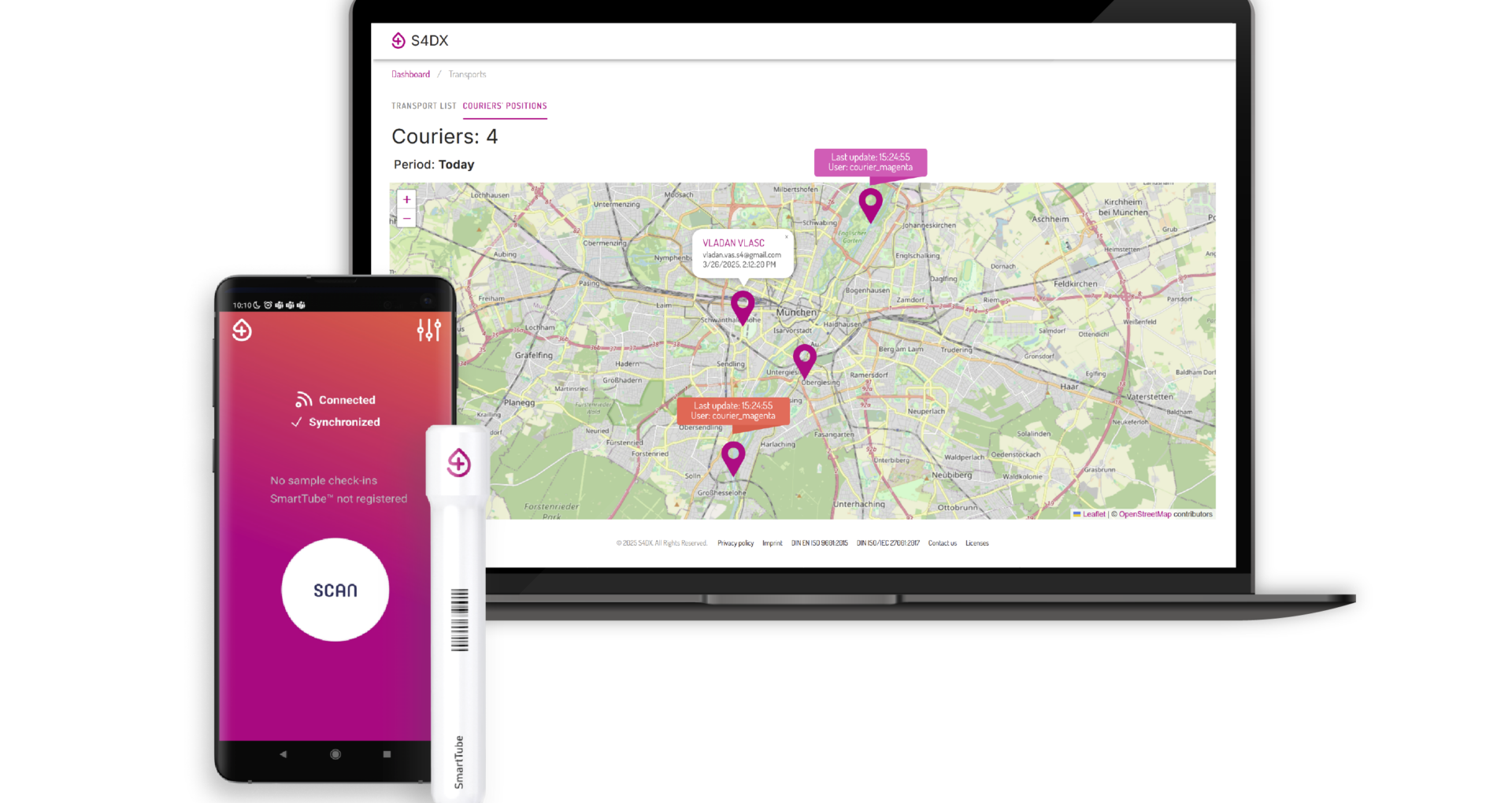
A Smarter Approach to Sample Transport
Technologies like S4DX SmartTube represent a new layer of assurance for labs aiming for ISO 15189:2022 alignment.
- These systems capture temperature, shock, and transport time data automatically, linking every event to the sample’s unique digital record.
- Information is made available in real-time, enabling transport teams to act while samples are still in transit — not hours later after delivery.
- During audits, full-chain documentation and alerts about any deviations are immediately accessible, making compliance easier and more transparent.
Building On What Works
Smart sample transport isn’t about discarding what has always worked in labs; it’s about adding consistency and the kind of actionable insight that auditors look for — and that patients deserve.
- With comprehensive digital records, lab managers can not only prove compliance, but also respond proactively to threats to sample integrity.
- Teams gain confidence, knowing that every step of the journey is monitored, documented, and ready for review at any time.
The Future of Chain of Custody
As regulatory expectations rise, so too must laboratory standards. Investing in smart transport tools helps labs stay ready — not just for the next audit, but for the demands of tomorrow’s clinical diagnostics landscape.